 DRUGS VERSUS COSMETICS
DRUGS VERSUS COSMETICS
First, it is important to discuss essential differences regarding what is considered a drug versus a cosmetic. The target focus and function for a drug may be beyond the scope of what may be claimed for cosmetics and personal care products. Pharmaceutics applied topically can intervene in the physiology of the skin and subjacent tissue with potential systemic affects with the metabolism of cells. Drugs may be intended to enter the blood capillaries in the dermis. They are used for skin treatment and prevention of disease. The purpose is to restore physiological functions or modify them in order to relieve the implications of a disease such as pain or inflammation or to prevent proliferation of a condition. Drug interaction is very individual depending upon how they are received metabolically within the cells. A medical professional carefully monitors them.
Cosmetic formulations also address variable skin conditions and support improvement to the appearance of the skin. The question that sometimes creates controversy is whether cosmetic ingredients can produce a pharmaceutical (physiological) effect without interfering with the composition of the epidermis. Lipids play an essential role in the stratum corneum organization and contribute to maintaining moisture balance. Innovative formulations containing physical structures and chemical composition similar to the skin’s defense layers tend to support the natural barrier without impairing the internal regeneration process in the skin.2 Earlier research by Albert M. Kligman found that simple moisturizers have clinical effects.3,4
There are numerous active agents such as vitamins A, C, and E esters, botanical extracts, trace elements, alpha-lipoic acid, curcumin, ceramides, linoleic acid, phosphatidylcholine and hyaluronic acid that have properties that support the skin. Ingredients must be formulated to work in synergy with one another and be able to be transdermally delivered respecting the pathways available through the skin barrier in the epidermis. A chosen pathway and penetration of active ingredients is dependent upon the molecular structure of a formula and delivery vehicle.
OVERVIEW OF THE STRATUM CORNEUM AND PATHWAYS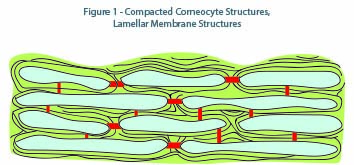
To understand cutaneous delivery of products, professionals must first understand the physiology of the skin and its complexities regarding the cells and their function. An essential purpose of the skin is to act as a barrier between the body and the surrounding environment. The acid mantle is the first line of defense of the skin, followed, secondly, by the stratum corneum. The skin provides a semi-permeable barrier to the transport of water and other substances. The stratum corneum also acts as a barrier to harmful substances that may attempt to enter the body.
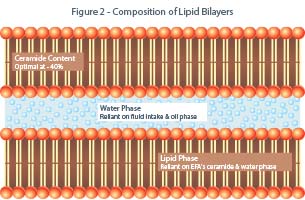
Often referred to as a brick and mortar structure, tightly compacted corneocyte structures are bi-laminar membranes (Figure 1) linked by corneodesmosomes tightly bound and separated by narrow intercellular spaces, forming a coherent membrane until they reach the upper surface where they progressively undergo a desquamation process.5 Lamellar structures (microstructures) are composed of fine, alternating layers of different materials in the form of lamellae. The adaptive capability of the stratum corneum regulates several key functions, including: permeability barrier formation, desquamation, maintenance of mechanical integrity, and generation of osmotically-active humectants (natural moisturizing factors). Composition of lipid bilayers consists of 30 to 50 percent ceramides, 25 percent cholesterol, and 10 to 20 percent free fatty acids (Figure 2). Moreover, it is also confirmed that the stratum corneum is an enriched environment of cytokines and can end up being an inflammatory tissue if its contents are extruded into the dermis, as in the case of acne pustules.6
 Topical compounds applied to the surface of the skin require a careful understanding of their purpose, how they are going to be delivered, and what response may take place within the microstructures of the skin once they leave the skin surface and move through the epidermal layers. How do they permeate in order to interact with skin cells and tissue? The skin barrier is governed by its components: 75 to 80 percent protein, 5 to 15 percent lipids, and 5 to 10 percent cholesterol and derivatives. The rate of transepidermal penetration of active ingredients depends upon the permeability of the focused area, as well as the molecular structure of the delivered compound. Active ingredients require enhancers in order to permeate the skin. Careful consideration is placed on how formulations will influence the site of application given that it incites structural changes within the stratum corneum. This includes potential triggering of the immune system. Moreover, is there a threshold of whether or not a chemical ingredient will trigger a harmful reaction?
Topical compounds applied to the surface of the skin require a careful understanding of their purpose, how they are going to be delivered, and what response may take place within the microstructures of the skin once they leave the skin surface and move through the epidermal layers. How do they permeate in order to interact with skin cells and tissue? The skin barrier is governed by its components: 75 to 80 percent protein, 5 to 15 percent lipids, and 5 to 10 percent cholesterol and derivatives. The rate of transepidermal penetration of active ingredients depends upon the permeability of the focused area, as well as the molecular structure of the delivered compound. Active ingredients require enhancers in order to permeate the skin. Careful consideration is placed on how formulations will influence the site of application given that it incites structural changes within the stratum corneum. This includes potential triggering of the immune system. Moreover, is there a threshold of whether or not a chemical ingredient will trigger a harmful reaction?
MECHANISMS FOR DELIVERY
Physical penetration into the skin can take place through the use of iontophoresis (polarizing) or sonophoresis (cavitation).Chemical transport occurs through specialized delivery systems placed in a formulation that serve as vehicles to carry active substances through the stratum corneum.
Enhancers include organic solvents, fatty acids, alcohols, surfactants, or property enhancers.7
The function of a delivery molecule is to increase permeability through a chosen pathway in order to bring active ingredients that will help improve the skin. Actives that simply remain on the surface of the skin will provide temporary benefit. Actives that permeate the stratum corneum layers work more efficiently to support a change in the appearance of the skin.8
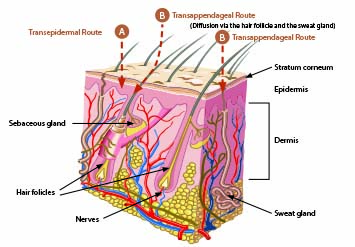
 Topically applied substances (drugs or cosmetic active ingredients) have three possibilities for permeating the skin: transcellular, intercellular, and intrafollicular (Figure 3).9,10 The delivery pathway and diffusion may vary depending upon the molecular makeup of a compound.
Topically applied substances (drugs or cosmetic active ingredients) have three possibilities for permeating the skin: transcellular, intercellular, and intrafollicular (Figure 3).9,10 The delivery pathway and diffusion may vary depending upon the molecular makeup of a compound.
Using a transcellular pathway, the topically-applied substance goes through the stratum corneum. With intercellular pathways, most compounds enter the skin between the stratum corneum corneocytes. In intrafollicular or transappendage pathways, penetration occurs through openings around hair follicles via the sebaceous glands and into sweat glands.
Examples of delivery enhancers include vesicular systems such as liposomes. They are microscopic and composed of amphiphilic (attracted to lipids) molecules made up of phospholipids that have the ability to mix with the stratum corneum lipid matrix. As they dissolve, they release their contents into the matrix and increase the stratum corneum lipid bilayer in the lamellae.11
Ceramides are a vital component of the stratum corneum that help repair and maintain the barrier. When applied in a specific concentration combined with fatty acids and cholesterol, ceramides have shown to improve skin barrier resistance.12 It is important that there is an understanding of the correlation between the chemical structures of the enhancer and how it interacts with the cells during its delivery. They must be formulated correctly so as to not create an imbalance.
Delivery vehicles such as liposomes and other similar, molecular-vesicle structures, not only support product diffusion, but they also protect active-encapsulated molecules, stabilizing them and preventing oxidation. This enhances the efficacy of the active.
 DELIVERY SYSTEMS
DELIVERY SYSTEMS
The following summary is a partial list of delivery systems used in skin care formulations. See the references for further reading. The terms lipophilic (lipid soluble) and amphiphilic (water soluble) may be useful in understanding these delivery systems.
Liposomes
Liposomes are hollow, fluid-like vesicles in spherical shape and contain lipid layers. They are the bilayer variant of nanoparticles and their structures are derived from natural cells. They can be used for encapsulating water-soluble active agents such as vitamin C. Once they enter the skin, they slowly release their contents.
Niosomes (nonionic)
Niosomes are non-ionic, surfactant-based vesicles. Similar to liposomes, they are used to deliver lipophilic drugs or cosmetic actives. They are the synthetic relatives of liposomes.
Microcapsules
Small, spherical, or oblong-shaped, microcapsules have an internal core that has an external cell or membrane. Multilayer spheres are capable of stabilizing both hydrophobic and hydrophilic actives, preventing oxidation and discoloration of the finished formula. They are used in topical cosmetics and personal care products.
Nanotechnology
This technology has been in existence since 1959. Nanotechnology is a delivery methodology into and through the skin by means of nanometer-sized particles. There has been some controversy around this technology since it is not always understood. In the skin care industry, two kinds of nanoparticles are used – non-biodegradable (solid) and biogradable (liquid). Biodegradable means the components of the nanoparticles are metabolized in the same way as other compounds contained in creams and lotions. Titanium dioxide is a component of solid nanoparticles. Fluid or liquid nanoparticles are oil bodies with lipid-soluble, active agents that are surrounded by a phosphatidylcholine (PC) membrane. Lecithin, fluid or liquid nanoparticles, are sometimes called nanosomes or nanodispersion. Other components include physiological ceramides, phytosterols, fatty acids, and vitamins.13
Microsponge
Microsponge technology is a patented, polymeric delivery system that is non-irritating, non-mutagenic, non-allergenic, non-toxic, and nonbiodegradable.They have the capability to release active ingredients in a time-released manner.
Hydrogels
Hydrogels are sustained-release delivery systems that are hydrophilic polymers. They offer a pathway of absorption and release of hydrophilic ingredients. Plants, seaweed, animal tissue, and chitin are examples of hydrogels found in nature.14 Hydrogels swell with a special type of diffusion referred to as imbibition.
Aspasomes
Ascorbyl palmitate was explored as a bilayer, vesicle-forming material. When combined with cholesterol, it formed vesicles called aspasomes. They have antioxidant properties. Aspasomes encapsulate hydrophilic ingredients to enhance skin permeation.15
TERMS AND REGULATIONS: pharmaceutical, cosmeceutical, over-the-counter, drugstore cosmetics
The difference between these categories is in the degree of actives found in them, as well as the conditions for which they are intended.16 The following information clarifies regulatory factors.
Drugs
The Food and Drug Administration (FDA) regulates and approves pharmaceutical, prescription drug manufacturers since they are producing products used for indications of disease. They are produced under quality control of purity that also requires clinical trials prior to being released for consumer use.
Cosmetics
The term pharmaceutical-grade or medical-grade used in some current marketing practices can be very misleading. The FDA does not require pre-approval for cosmetic products and ingredients, with the exception of color additives. Instead, the FDA encourages cosmetic manufacturers to participate in FDA Voluntary Cosmetic Registration programs for cosmetics, and also recommends the possible use of the International Organization for Standardization (ISO) and Good Manufacturing Practices (GMP). The FDA can inspect facilities to assure product safety compliance with the GMP. Moreover, they will take regulatory action towards adulterated and misbranded cosmetics. The FDA regulates the Fair Packaging and Labeling ACT.17
 Over-the-Counter Drugstore Products
Over-the-Counter Drugstore Products
There is indeed a market for products found in drugstore and department store venues. They address general skin concerns and are marketed to a mass consumer base. Due to this higher market, products may contain lower concentrations or almost no actives. This is for consumer safety.
There is also an issue of self-diagnosing. Many consumers do not ask for professional help when encountering skin issues. Chronic skin conditions may require treatment with a licensed professional, including medical diagnosing with explicit direction from a physician who prescribes a pharmaceutical product with the strongest level of actives. The higher the actives, the more caution required, including regulating usage and monitoring potential side effects.
Cosmeceuticals
The cosmetic industry uses the term cosmeceutical for cosmetic products that may have medicinal or drug-like benefits. According to the Federal Food, Drug, and Cosmetic (FD&C) Act, drugs mitigate or prevent diseases or affect the structure or function of the human body. The term cosmeceutical has no meaning under the laws of the FDA.18 A product can be a drug, a cosmetic, or both depending upon claims and clearances. That said, there are hundreds of cosmetic formulations and ingredients that may extend restorative affects to the skin without interfering with the physiology of the living epidermis. There may be skin improvement for skin barrier disorders such as dry skin, for example, from simple ingredients like phosphatidylcholine (from shea butter) or other skin-mimetic components free of counterproductive additives. These counterproductive additives can include perfumes, allergenic preservatives, non-degradable emulsifiers, or comedogenic hydrocarbons, as well as other questionable substances.19
Bioactive ingredients found in cosmeceuticals have benefits that are higher than over-the-counter brands of skin care. They may have specific formulations to address issues such as acne, aging and wrinkled skin, inflammation, rosacea, and barrier repair. Some of these include vitamins, peptides, trace elements (copper peptides for collagen formation), advanced glycation end (AGE) inhibitors, retinoids, and phosphatidylcholine. They are normally found in a medical environment or in a licensed, experienced skin care facility.
Regulation
There are regulatory agencies in North America (the FDA and United States and Canada Health) and in Europe and Asia. Based in Washington, D.C., the Personal Care Products Council – formerly the Cosmetic, Toiletry, and Fragrance Association (CTFA) – represents the global cosmetic and personal care products industry. They help standardize and establish rigid guidelines for safe manufacturing, quality control, and safety standards for raw materials, consumer safety, truth in labeling, testing and clinical trials, and a host of other parameters.20 The International Cosmetic Ingredient Nomenclature Committee (INC) is charged with the responsibility of designating INCL names to identify cosmetic ingredients. INCL names are unified worldwide and must be consistent on all labels.
Cosmetic formulations and delivery mechanisms are very complex. In order to be efficacious, they must be compatible with the skin. Professionals must understand the science of the skin and its function, as well as the disorders that may occur with clients, especially as they age and undergo life changes. Bringing the right product to the skin requires study of what components are going to best serve clients’ skin.
Resources
1 Lautenschlager, Hans, Ph.D. Medical Beauty Forum. 2014. no. 4, 16-18.
2 Barrett-Hill, Florence. “Emulsifiers in Skin Care: Purposes, Properties and Disadvantages.” International Association for Applied Corneotherapy. 2017. https://corneotherapy.org/articles/226-emulsifiers-inskin-care.
3 Kligman, Albert M. “Corneobiology and Corneotherapy - a Final Chapter.” International Journal of Cosmetic Science 33, no. 3 (2011): 197-209. doi:10.1111/j.1468-2494.2011.00644.x.
4 Bouwstra, J., PL Honeywell-Nguyen, GS Gooris, and M. Ponec. “Structure of the Skin Barrier” in Skin Barrier, ed. Peter Elias and Kenneth Feingold (NY, London: Taylor and Francis, 2006), 75-76.
5 Kligman, Albert M. “How the Dead Stratum Corneum Became Alive” in Skin Barrier, ed. Peter Elias and Kenneth Feingold (NY, London: Taylor and Francis, 2011), 15.
6 Kligman, Skin Barrier, 197-209.
7 Rosen, Meyer and Ameann DeJohn. “Intelligent Delivery Systems for Enhancing the Performance of Active Ingredients in Skin Care Formulations.” Experts. https://www.experts.com/Articles/Intelligent-Delivery-Systems-Active-Ingredients-Skin-
Care-Formulations-By-Meyer-Rosen
8 Rosen, “Intelligent Delivery Systems.”
9 Tran, Thanh-Nga T. “Cutaneous Drug Delivery: An Update.” Journal of Investigative Dermatology Symposium Proceedings 16, no. 1 (2013): S67-S69. doi:10.1038/jidsymp.2013.28
10 Dayan, Nava. “Pathways for Skin Penetration.” Cosmetics & Toiletries Magazine, June 2005, vol. 120.
11 Dayan, 74.
12 Dayan.
13 Lautenschlager, Hans, PhD. “Nanoparticles in cosmetic products – good or bad?” Beauty Forum, no. 5 (2009): 44-47.
14 Rosen, “Intelligent Delivery Systems.”
15 Gopinath, D., Ravi D, Rao BR, Apte SS, Renuka D, and Rambhau Det. “Ascorbyl palmitate vesicles (Asapasomes): formation, characterization and applications.” International Journal of Pharmaceutics, 271, no. 1-2 (2004): 95-113. https://www.ncbi.nlm.nih.gov/pubmed/15129977
16 Skelly, Michelle. “Key Differences between Drugstore, Cosmeceutical and Pharmaceutical Skin Care.” 2016. https://www.linkedin.com/pulse/key-differences-between-drugstore-cosmeceuticalskin-care-skelly
17 “FDA authority Over Cosmetics: How Cosmetics are not FDA-Approved, but are FDA-Regulated.” https://www.fda.gov/Cosmetics/GuidanceRegulation/LawsRegulations/ucm074162.htm
18 “U.S. Food & Drug, Cosmeceutical.” https://www.fda.gov/Cosmetics/Labeling/Claims/ucm127064.htm
19 Barrett-Hill, “Emulsifiers in Skin Care.”
20 Daum, Casey Mee Lee. “Self-Regulation in the Cosmetic Industry: A Necessary Reality or a Cosmetic Illusion?” 2006.
https://dash.harvard.edu/bitstream/handle/1/8965615/Daum06.html
 Alexandra J. Zani is an international educator, researcher, and author with a background in cell biology. Her passion for education resulted in receiving numerous advanced certifications, both in the United States and abroad. Zani earned an instructor license for aesthetics and cosmetology, is NCEA certified, and is certified in Oncology Esthetics® and the Pastiche Method® of Skin Analysis. She is a member of the International Association for Applied Corneotherapy (IAC). Zani presents education for advanced aesthetic technology, including microcurrents, LED, and non-ablative laser. She is a specialist in anti-aging sciences including the effects of nutrition, lifestyle, and the mind and body connection.
Alexandra J. Zani is an international educator, researcher, and author with a background in cell biology. Her passion for education resulted in receiving numerous advanced certifications, both in the United States and abroad. Zani earned an instructor license for aesthetics and cosmetology, is NCEA certified, and is certified in Oncology Esthetics® and the Pastiche Method® of Skin Analysis. She is a member of the International Association for Applied Corneotherapy (IAC). Zani presents education for advanced aesthetic technology, including microcurrents, LED, and non-ablative laser. She is a specialist in anti-aging sciences including the effects of nutrition, lifestyle, and the mind and body connection.
Want to read more?
Subscribe to one of our monthly plans to continue reading this article.