Syneron Medical Ltd., a global market leader in the aesthetic medical device marketplace, recently announced that it has received the United States Food and Drug Administration (FDA) 510(k) clearance to market the UltraShape™ System for non-invasive reduction of abdominal circumference via fat cell destruction.
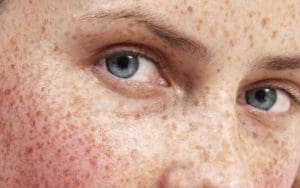
New Advances in Treating Hyperpigmentation
This past decade has seen an enormous surge in popularity of skin brightening products formulated specifically to treat hyperpigmentation and…















0 Comments