Most chemists openly express their concern over how little time is spent studying surfactants in college given their considerable importance in so many industries, especially the skin care industry. According to Perry Romanowski, coauthor of the acclaimed textbook, Beginning Cosmetic Chemistry, “surfactants are the workhorses of the cosmetic industry and all cosmetic chemists would benefit learning the basic principles of surfactant chemistry.” 1 Most often, action ingredients take all the glory for product performance, while the surfactant’s contribution is overlooked and frequently misunderstood by the consumer.
In some instances, their safety on humans and in the environment has even been questioned and scrutinized. Almost everything we use and work with in skin care treatments is based on the science of surfactants and emulsifiers. Soaps and cleansing agents are emulsifiers and surfactants. The majority of creams and lotions are emulsions. The terms surfactant and emulsifier are often used interchangeably; however, there is a fine line between the two, which can be industry specific. Food chemists use emulsifier more, where industrial chemists use the term surfactant more. Cosmetic chemists make the most prominent distinction between the two categories of ingredients, as the action of a surfactant is perceived very differently than that of an emulsifier in skin care formulations. A common misconception is to only think of ingredients that lather and foam as surfactants, when basically, surfactants are ingredients that bring oil and water together. That being said, it is important to recognize that emulsifiers are a group of chemicals belonging to the surfactant family.
Surfactants
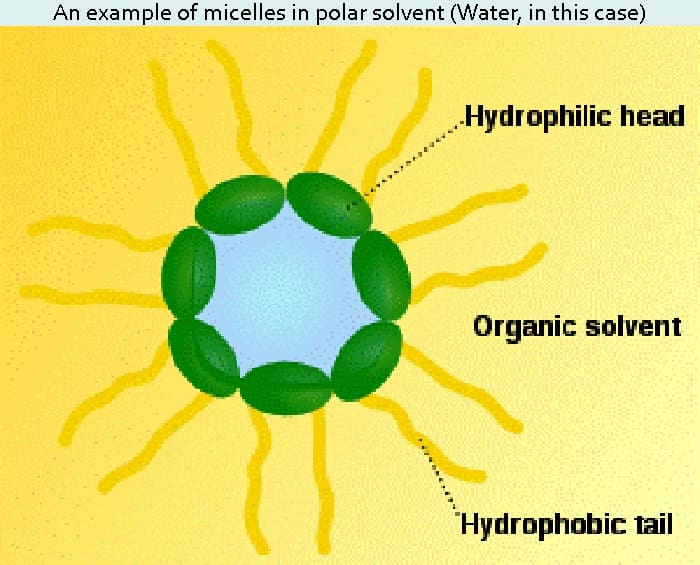
Surfactants are any ingredients that lower tension between a surface and a liquid or between two or more immiscible substances. Different types of surfactants are found in facial cleansers, soaps, body washes, shampoos and emulsions. Surfactants work by becoming imbedded in both oil and water substances. They do this by having molecules with an oil-loving tail and a water-loving head. Surfactants are some of the most versatile ingredients in skin care, playing numerous roles in cosmetic formulations. They can act as detergents for cleansing, wetting agents for better spreadability, foaming agents to produce luxurious suds, emulsifiers to create stable mixtures of oil and water, conditioning agents to improve the appearance of hair and skin, solubilizers to help mix fragrances and essential oils into water-based formulas, preservatives to keep cosmetics microbe-free, and special effects to improve the look of certain formulas. 2 Some examples of surfactants are sodium or ammonium lauryl or laureth sulphate, sodium methyl cocoyl taurate, sodium lauroyl or cocoyl sarcosinate cocomidopropyl betaine, triethanolamine (TEA) compounds, dethanolamine (DEA) compounds, monethanolamine (MEA) compounds, polyethylene glycol (PEG) compounds, Quaternium -7, 15, 31, 60, et cetera, lauryl or cocoyl sarcosine disodium oleamide or dioctyl sulfosuccinate.
Surfactants as Cleansing Agents
The most common function of surfactants is to cleanse. Many consumers still believe that the amount of foam a cleanser produces is an indication of its cleansing efficiency, when, in fact, the amount of foam has much more to do with the aesthetics of the product, rather than its cleansing potential. Ingredient names containing the words sulfate, sulfocuccinate, or the prefix sarco, indicate strong cleansing tendencies and provide luxurious, thick foam. Sodium lauryl sulfate, sodium laureth sulfate, disodium lauryl sulfosuccinate, cocoyl sarcosinate are all representative of this group. Milder alternatives to these ingredients that contain words in their names like ampho or the suffix -taine, such as cocoamphocarboxyglycinate and cocobetaine, are more suitable for barrier-compromised skin that cannot tolerate strong surfactants.
In recent years, the sulfate-free claim has become one of the most popular marketing claims, promoting low-foaming cleansing options as healthier, barrier-friendly alternatives to high foaming, potentially dehydrating cleansers. Likewise, consumer demand for surfactants from sources other than petrochemical has contributed to the development of greener alternatives, some of which are derived from innovative sources such as cannabis sativa seed oil PEG-8 Esters. Other green surfactants include ingredients from oats, apples, coconut, sugar beets and sweet almond seeds, such as sodium lauryl oat amino acid, sodium cocoyl glutamate, sodium cocoyl hydrolyzed, amaranth protein, disodium sulfosuccinate laurylglucoside crosspolymer, potassium olivoyl hydrolyzed oat protein, sodium cocoyl apple amino acids, sodium, sweetalmondamphoacetate, saponins, and betaine.
Emulsions and Emulsifiers
An emulsion can be defined simply as two immiscible fluids in which one liquid is dispersed as fine droplets into the other. Homogenized milk is an example of a typical oil-in-water (o/w) emulsion. Milk fat, representing oil phase, is dispersed in water by the homogenization process. Emulsifiers, in this case a milk protein called sodium caseinate, as well as several phospholipids, prevent the fat and water from separating. Micelle is the term frequently mentioned when emulsions are discussed. It refers to the way water-loving heads and oil-loving tails arrange themselves in a special way, depending on the environment. They are usually driven to arrange either with the water-loving heads out and the oil-loving tails in (o/w) or with the water-loving heads in and the oil-loving tails out (w/o).
The most common emulsions in the cosmetic industry (about 80 percent) are the ones in which oil is dispersed in water (o/w). They are high water content and, therefore, low in cost. These emulsions are favored for their stability and flexibility, as well as their non-greasy, non-oily feel. On the other hand, w/o systems offer longer lasting emolliency, wash-off resistance, and barrier protection. They spread more easily on skin, and often leave a lubricious, rich or oily feel, favored by mature clients and consumers with dry, barrier-compromised skin. These formulas tend to be less cost effective, as oils are always more costly than water and these types of emulsions are generally more difficult to produce.
To create emulsions, typically a surfactant (emulsifier) is used. Emulsifiers blend and hold together ingredients that would not normally mix very well. In cleansing products, the ability of an emulsifier to combine waters and oils gives a final product the ability to both cleanse and condition the skin at the same time. Some commonly used emulsifiers in skin care are glyceryl stearate, PEG-100 stearate, stearyl alcohol, cetyl alcohol, laureth-23, steareth alcohol, cetyl/PEG/PPG 10 dimethicone, and stearic acid.
Fatty acids are key components of many cosmetic emulsifiers, due to their miscibility in a variety of natural and synthetic oils. They are derived from natural oils such as coconut, palm kernel, sunflower, wheat germ, and so on. They are used as conditioning agents to improve the surfaces of hair and skin, thickening agents to make thin products creamier, as secondary emulsifiers to help create stable mixtures of oil and water, and as opacifying agents to make formulas look more luxurious.
Emulsifiers have a hydrophilic or water-loving head and a lipophilic or fat loving tail. The hydrophilic head clings onto the water phase of an emulsion and the lipophilic tail creates a ball around the oil-based ingredients, the oil phase of the emulsion.
Being that close to 80 percent of all skin care ingredients are at least in part derived from petroleum, it should come as no surprise that a great number of emulsifiers are petroleum/hydrocarbon derived as well. Esters, like polyethylene glycol or ethylene glycol, are called PEG esters and represent the most prominent group of petroleum-related emulsifiers. These emulsifiers make the creation of many beautiful, stable and unique emulsions possible, yet they are constantly under the scrutiny of the consumer. They are often called ethoxylated surfactants, as ethylene or propylene oxide is utilized in the chemical reaction. PEGs are either used alone or linked to other ingredients to enhance solubility into a product. Depending on the manufacturing process, PEGs may be contaminated with measurable amounts of ethylene oxide (a known carcinogen) and 1,4-dioxane (a possible carcinogen). PEGs are still being reviewed for genotoxic effects that can lead to mutations and cancer, but to date, nothing has been found. Chemists are actively researching alternatives, but the only known alternatives are either three to five times the cost of the current emulsification system or do not provide long-term stability. This is an ongoing issue for cosmetic chemists and product formulators.
Choosing an emulsifier is not only crucial for the stability of an emulsion, but can also have a large impact on formula consistency, skin feel, and perceived performance. The skin care market continues to be fast-paced and highly competitive. Consumers expect performance, convenience and superior aesthetics, along with long-lasting, highly efficient moisturizers and age-fighting creams that go on smoothly, without tackiness or residue. Innovative, multifunctional, high performance products have the best chance of success. Silicone emulsifiers and silicone emulsions are emerging as an additional option that can help bridge the gap between the o/w and w/o systems, providing the best of both worlds, helping to produce stable and aesthetically superior emulsions with high water levels, making them relatively affordable. These evolving surfactant and emulsion technologies are, and will continue to be, at the core of innovative formulations, fully capable of satisfying even the most demanding consumer requirements.
References
1 Randy Schueller & Perry Romanowski. Beginning Cosmetic Chemistry, 2nd edition. (2003) Allured Publishing Corporation. 2 Useful information about cosmetic chemistry http://thebeautybrains.com by Perry Romanowski , March 15, 2014 http://www.chemistryexplained.com/Co-Di/Cosmetic-Chemistry.html#b#ixzz2vz6UdelT Key Centre for Polymer Colloids. University of Sydney, NSW Australia http://www.hsc.csu.edu.au/senior_science/core/life_chem/9-2-2/9.2.2.html 9.2 Lifestyle chemistry: 2. Colloids and surfactants Reproduced with permission from NSW HSC Online http://hsc.csu.edu.au © NSW Department of Education and Communities, and Charles Sturt University, 2014 http://thebeautybrains.com by Randy Schueller , March 15, 2014 New Formulating Options with Silicone Emulsifiers. Isabelle Van Reeth Marilena Morè Dow Corning Europe. Robin Hickerson Dow Corning USA ©2003, 2009, 2012, Dow Corning Corporation



































0 Comments