Undoubtedly, as the global population continues to age, and years of sun damage, compounded by the influence of fluctuating hormones, manifests itself in uneven pigmentation, this category will continue to increase worldwide. Market research studies indicate that an uneven skin tone (as in hyperpigmentation and hypopigmentation) is perceived as “aged skin” while a more even skin coloration is judged to be “healthier and younger”. Today’s consumer is not only looking to treat age spots or hyperpigmentation, but to create a more even skin tone with enhanced luminosity.
Consumer demand for brightening products both here and abroad has spurred both the pharmaceutical and cosmetic companies to research and develop new technology and products that are not only effective but safe to use. As professional skin care therapists, our focus must also be on seeking out products that are safe and effective for brightening the skin without causing irritation or damage, which can lead to premature aging or worse.
What determines skin color?
Skin color is mainly determined by the amount of melanin present in the skin. Constitutive skin color is the basic melanin content of our skin that is genetically inherited without any influence from sun exposure or hormones. Facultative skin color is the result of other influences, such as sunlight and hormones, and their impact on our skin color. When we discuss hyperpigmentation, we are referring to facultative skin color.
Melanin, the brown or reddish skin pigment, is synthesized in dendritic cells called melanocytes. Regardless of race, we all have approximately 800 to 1,000 melanocytes per square millimeter of epidermis. Melanocyte cells look like an octopus with long arms or dendrites extending upward into the stratum spinosum layer. Melanocytes in sun exposed skin are larger with branched dendritic arms when compared to those found in protected skin. When exposed to UV light, the pituitary gland in the brain generates a melanotropin called alpha- or beta-Melanin Stimulating Hormone (alpha- or beta-MSH). This melanotropin molecule binds to a specific receptor site (MC1-R) on the melanocyte signaling melanin formation to commence. (This receptor site is also affected by hormones such as estrogen, which accounts for the influence that hormones have on melanin formation.) Alpha-MSH not only stimulates pigment formation but it also triggers the inflammatory and immune response. Likewise, inflammatory modulators that trigger the immune response also trigger melanogenesis. Hence, what was once called an epidermal-melanin unit is now more accurately called the keratinocyte- langerhans-melanocyte complex.
Once the melanocyte is activated, eumelanin (black melanin) formation commences with activation of a key enzyme, tyrosinase. We will discuss this in more depth in the section entitled “How is melanin made?”
Melanin pigment is bound to a protein matrix and contained in an oval-shaped structure known as a melanosome. After the melanosome is produced in the melanocyte, it is transferred to a neighboring keratinocyte cell where it begins its journey through the different layers of epidermis while contributing to the skin’s color.
Regardless of skin color, the number of melanocytes does not vary among humans. There is approximately one melanocyte for every 36 keratinocytes found in the epidermis. The amount of melanin, the type of melanin produced (whether it is eumelanin, a brownish-black pigment or phaeomelanin, a yellowish-red pigment), the size of the melanosomes and the distribution of melanosomes in the epidermis all contribute to the skin's color and intensity – in other words, the lightness or darkness of the skin. In black skin, we not only see larger melanosomes with more melanin present, but more of them. They are seen as larger, individual, jelly bean-like structures surrounded by a membrane, whereas melanosomes in Caucasian, Asian and Hispanic skin, are smaller, vary in size and shape, and have many melanosomes clustered in a single membrane jacket.
While the amount of melanin synthesized in our skin is determined by our genetics (constitutive skin color), there is also an overriding affect of the environment, such as exposure to UV radiation, that determines skin pigmentation levels (facultative color). Due to its ability to absorb UV radiation, the primary function of melanin is believed to be to protect the cells/skin from sunlight. Upon exposure to UV radiation melanin synthesis is stimulated resulting in a characteristic tan. Studies have shown that exposure to UV causes melanosomes to actually cluster, forming a protective cap over the nucleus of the cell. In addition to the development of an overall tan, exposure to UV light may also stimulate hyperpigmentation in specific areas such as the hands, face, and neck. These dark spots are often referred to as age spots or liver spots (lentigines), and are a nuisance for fair-skinned and darker complexions as well. Age spots usually become evident in our early 40s and become increasingly more evident as we age. By the time we reach 60 years old, 90 percent of all individuals will have sun-induced age spots. Lentigines are harmless, flat, brown discolorations of the skin, which usually occur on the back of the hands, neck, and face of people older than 40 years of age. Size can range from 2mm to 30mm. These spots, more than anything else, give away a person's age. Sun spots are caused by the skin being exposed to the sun over many years and unlike freckles, lentigines do not fade in winter. Freckles, also known as ephilides, are also flat spots that are red or brown and typically appear during the sunny months and fade in winter. They are most often found in individuals with fair skin and are generally a genetic trait. Both freckles (ephilides) and age spots (lentigines) look very similar to the naked eye. Short of monitoring their color intensity in the summer versus winter months, the only way to distinguish the two is to look at a section of the skin under the microscope. Lentigines display flattened rete ridges (the finger-like projections found at the interface between the epidermis and the dermis) and have noticeable pigmentation in the stratum basale layer. There are also more melanocytes in the region. Freckles do not show any change to the rete ridges and have a normal population of melanocyte cells.
Because light colored skin has less melanosomes, less melanin, and lower levels of eumelanin than darker skin, there is less protection against exposure to UV radiation. Likewise, in individuals with lighter skin, the majority of the melanin is confined to the lower layers of the epidermis; in darker skin, the melanin is evident throughout the layers of the epidermis with more melanin in the outermost layers of the stratum corneum. It is believed that the enzymatic process, whereby the usual protein/melanin complex is broken down as it transitions through the epidermis during keratinization, is not as active in dark skin. This is most likely part of the protective role that melanin plays in the skin. While it stands to reason that darker skin would be easier to treat due to the melanin being closer to the surface of the skin, the overall abundance of melanin in dark skin actually makes it more difficult to treat. Do not think for one minute that dark skin with its preponderance of melanin is exempt from the ravages of UV exposure. It may be better equipped to protect itself but it is still vulnerable to sun-induced hyperpigmentation as well as post-inflammatory hyperpigmentation, photodamage, and skin cancer.
Other causes of pigmentation:
Post-inflammatory hyperpigmentation – In addition to genetics and environmental influences, skin pigmentation is affected by endocrine or hormonal factors, usage of prescription drugs, stress, topically applied products including cosmetics, and wound healing to the skin tissue. The latter incidence gives rise to post-inflammatory hyperpigmentation (PIH), a phenomenon that is more problematic for individuals with darker skin color. PIH stems from the melanocytes exaggerated response to cutaneous insult, which results in an increased or abnormal distribution of melanin in the tissues. Interestingly, melanocyte activity is stimulated by the same inflammatory mediators that are activated when the skin’s immune response is activated. What affects the skin’s Langerhans cells will generally stimulate the melanocytes, and vice versa. When inflammation subsides, the inflammatory mediators revert to normal levels and so does the production of melanin. In due course, the cells causing hyperpigmentation rise to the stratum corneum and slough-off, causing the hyperpigmentation to disappear. Recent studies have shown however, that depending on the depth of the inflammation or wound, hyperpigmentation can not only be evident in epidermal cells but in the dermis as well. This may very well account for the difficulty in treating post-inflammatory pigmentation associated with deeper scar tissue.
Melasma – Hormonally induced pigmentation manifests itself in various forms, such as hyperpigmentation spots and melasma, better known as the mask of pregnancy.
Melasma affects primarily women of child-bearing age. Some studies suggest that up to 75 percent of women may develop melasma during pregnancy and about 33 percent of women on birth control pills. It is more prevalent in women with darker skin color and it is most commonly seen in areas that are exposed to sunlight. Some scientists say UV exposure seems to be a requirement for melasma to develop which may account for the fact that it is less noticeable in winter months when UV exposure is lower. It is seen as an irregularly shaped patch of hyperpigmentation ranging from light brown to dark brown. The most common pattern is the centrofacial – on the chin, upper lip, cheeks, nose, and forehead; to a lesser extent it is seen as malar: cheeks and nose. Even less common is the mandibular, jaw line pigmentation.
The name, mask of pregnancy, points to its frequent occurrence during pregnancy or when oral contraceptives are taken. It is thought that estrogen and UV light seem to be the biggest culprits in this condition. The hyperpigmentation may be permanent or disappear and reappear with subsequent pregnancies or use of the contraceptive pill. Although melasma is evident during pregnancy, it can also appear as the result of some ovarian disorders. It is believed that both estrogen and progesterone influence melanocyte activity, driving the production of melanin. When compared to melanocyte activity that is stimulated by sun exposure, hormonally induced melanocytes are considered to be hyperactive. Hormonally induced melanin may be confined to the epidermis, but it may also be found in the dermis making treatment especially difficult. Once hormonal fluctuations subside, such as the end of pregnancy, or discontinuance of hormone supplements or birth control pills, the hyperpigmentation often disappears. However, the condition may persist up to five years after pregnancy or stopping birth control pills. And unfortunately, once a person has it, they are more prone to get it again. To complicate matters, the melanin may reside in both the dermis and epidermis, which accounts for why it is so difficult to treat (treating the dermal component is almost impossible with topically applied products).
How is melanin made?
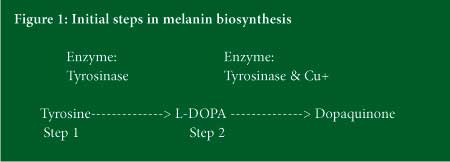
In order to understand how we can effectively treat hyperpigmentation, we need to review how melanin is made in the skin. The process of melanin production is known as melanogenesis. The production of melanin by the melanocyte is a complicated biosynthetic pathway which starts with a key amino acid, tyrosine. There are many chemical reactions that take place, some involving co-factors that must be present in order for the reactions to occur. The most critical steps in the biosynthesis of melanin appear to be the first two steps that are mediated by the enzyme tyrosinase, a copper containing enzyme that catalyzes two distinct reactions in the production of melanin. Up until very recently, most of the ingredients that we use to fight pigmentation affect these first two steps in melanin synthesis.
The production of pigment within the melanocyte is a multi-step process. The first step, mediated by the enzyme tyrosinase, involves the conversion of the amino acid tyrosine to L-DOPA. In the second step, L-DOPA is converted to dopaquinone, a dihydroxybenzene derivative; this step is also mediated by the tyrosinase enzyme but it now requires copper as an enzyme co-factor. Once Dopaquinone is made, the biosynthetic pathway splits and several subsequent reactions lead to either eumelanin, the brown and black melanin, or phaeomelanin, the yellow and red melanin pigments. Scientists are continually studying this biosynthetic pathway in an effort to better understand and control the process of melanin formation.
Eumelanin is the brown/black pigment that we find in Caucasian, Asian, and black skin. This is the most common pigment that we see in the skin. The content and intensity of this pigment will give an indication of the degree of photo protection.
Pheomelanin is the yellow/red pigment that we see in fair skin and red hair. It is synthesized from tyrosine and the amino acid cystine, which means it is less stable to UV light. Consequently, pheomelanins oxidize more readily which explains why red hair is relatively rare. This type of melanin offers no photo protection to the skin.
How can we control hyperpigmentation?
The most obvious means of controlling pigmentation, especially sun-induced, would be to apply a broad-spectrum sunscreen with a minimum SPF of 15 regularly. This will help control future pigmentation.
What about existing hyperpigmentation? For years hydroquinone has been considered to be the most effective ingredient for lightening hyperpigmentation. It is classified as an over-the-counterdrug in the U.S. and may be used in concentrations up to two percent. There are various theories on how hydroquinone works to affect hyperpigmentation; some researchers claim that it denatures the melanin-protein complex, thereby causing a discoloration of skin. Others claim it inhibits the Tyrosinase enzyme, while some claim it inhibits the synthesis of the protein associated with the melanin. While hydroquinone may be an effective pigment lightener, there are many concerns regarding its safety. It has been estimated that one third of the population is allergic to hydroquinone. Serious contact dermatitis has been noted and skin may be photosensitized with prolonged use. Hydroquinone has been classified as "an extreme sensitizer". In some cases, a condition known as onchronosis can result, leading to actual hyperpigmentation and acne-like lesions. Others report that hypopigmentation results when used on an olive skin color. The Occupational Safety and Health Administration in the U.S. states that hydroquinone is, “mutagenic and has cancer causing potential”. “The Addendum to the Final Report on the Safety Assessment of Hydroquinone” published in the Journal of the American College of Toxicology in 1994 concludes that hydroquinone is a potent cytotoxic agent that causes mutations and alterations to DNA and that it should not be used in any leave-on type of product; it is safe for rinse-off products when used in concentrations less than one percent. Remember, most OTC products marketed to lighten pigmentation use hydroquinone at a two percent concentration. Bearing in mind the results of these studies, it should really come as no surprise that the use of hydroquinone has been banned in many countries throughout the world.
Hydroquinone is the only ingredient recognized as a lightening agent by the U.S. FDA. Consequently, any other ingredient used to lighten skin must be referred to as a brightening agent and not a lightener. In the past few years, as hydroquinone has fallen out of favor and even banned in most of the world, there has been an onslaught of alternative brightening agents that are now marketed to combat hyperpigmentation.
In 2007 the U.S. FDA reported that it intended on banning the use of hydroquinone in non-prescription products (due to safety issues) but as of yet, they have not implemented any new regulations.
Safe Alternatives for Brightening Skin
Studies have shown that we can control the process by influencing various steps along the pathway. Perhaps the best-studied step involves that of the tyrosinase enzyme. For years, scientists have been looking for ways to regulate this enzyme and therefore, slow melanin formation. One can regulate tyrosinase by either slowing down its activity or competing for its substrates, either Tyrosine in step one (see Figure 1) or L-DOPA in step two. An example of a mechanism that controls tyrosinase activity would be the use of rice extract – the phytic acid found in rice binds the copper metal ion, slowing down step two of the tyrosinase mediated reaction. Kojic acid, Aspergillus (a fungus) ferment, Rumex extract, and ergothioneine (a plant amino acid) are other examples of tyrosinase inhibitors that chelate or bind copper. Newer ingredients, such as hydroxycinnamic acid, gluconic acid, and zinc glycinate also chelate copper to control tyrosinase activity. Of particular interest is zinc glycinate, which stimulates synthesis of an antioxidant protein called mettallothionein that binds the copper stopping step two in the pathway. In addition it has been shown to reduce tyrosinase synthesis and activity, as well as suppress melanocyte growth factors that stimulate melanin synthesis.
Many botanical extracts have been studied for their ability to inhibit tyrosinase. These include: Sophora angustifolia, Kiwi fruit, Nasturtium, Rumex (Yellow Dock), Phyllanthus emblica fruit, Mulberry, Bearberry, Licorice, and more recently Ferula foetida (giant fennel). While the precise mechanism of how these work is not clearly understood, studies have been conducted looking at the flavonoid components of these plants and their similarity in chemical structure to L-DOPA. These flavonoids are dihydroxybenzene derivatives, which are thought to compete with the substrate in either of the tyrosinase mediated steps. Many of these botanical extracts may in fact work on two fronts – not only do they inhibit the tyrosinase enzyme, but they compete with the enzyme’s substrate as well.
A different approach to controlling pigmentation is illustrated with the use of yeast extract, which has been studied for its ability to shunt melanogenesis toward the lighter phaeomelanins. This gives the perception of a lighter pigmentation, although actual production of melanin is really not turned off.
While the use of hydroxy acids (lactic acid, glycolic acid, and salicylic acid) in skin brightening products has generally been utilized to accelerate desquamation and removal of pigmented keratinocytes, it has recently been shown that a five percent concentration of lactic acid will inhibit the formation of the tyrosinase enzyme, thereby slowing the process of melanin synthesis. This new approach to affecting melanin synthesis is unique to lactic acid and does not occur with other alpha hydroxy acids such as glycolic acid or the beta hydroxy acid, salicylic acid. Other exfoliating agents used in brightening products include pumpkin enzyme, sutilains (a protease enzyme), lactobacillus ferment, and galactoarabian, a molecule that stimulates natural desquamation in the skin.
Kojic acid at up to one percent concentration has also been used to treat hyperpigmentation; its ability to bind the copper metal and inhibit tyrosinase enzyme accounts for its effectiveness. However, best results are obtained in an anhydrous base (no water), which is not generally how it is marketed. Recent studies on kojic acid show that topical application may induce contact dermatitis and that it has a high sensitizing potential; hence it is banned in some countries.
Another means to control hyperpigmentation is to control inflammation. The use of anti-inflammatory agents such as licorice, green tea, white tea, and mulberry will help address the connection between inflammation and pigment formation. These extracts also may act as antioxidants slowing many of the oxidation steps involved in melanogenesis.
More recently scientists have been studying the use of other agents to control melanin formation. Niacinamide has been shown to stop the transfer of melanosomes to neighboring keratinocytes. Others agents, such as glucosamine and dithiooctanediol, have been shown to stop the activation of the tyrosinase enzyme, a step that involves glycosylation or the addition of a sugar molecule to the inactive pro-enzyme structure converting it to the activated enzyme. If the enzyme remains inactive, melanin formation ceases. At the American Academy of Dermatology meeting, held in 2006 a group of scientists reported superior lightening effects on hyperpigmentation when niacinamide was combined with glucosamine.
Newer research indicates that we can also control melanin formation by affecting the signaling process involved in melanin biosynthesis. We can impact the messengers that signal melanin synthesis by using sunscreens and anti-inflammatory agents. Newer, state–of-the-art ingredients such as Ascophyllum nodosum (a brown seaweed extract), has been shown to inhibit endothelin-1 (ET-1), a molecule synthesized and released from the keratinocytes after UV exposure. ET-1 stimulates melanocyte proliferation and dendritic length, as well as tyrosinase activity in melanocytes. By inhibiting the signal molecule ET-1, we can therefore inhibit melanin formation. Likewise, the use of Palmaria palmata, a red algae, has been shown to inhibit the release of Stem Cell Factor (SCF), another signaling molecule released by keratinocytes upon exposure to UVB radiation. SCF binds with a receptor on the melanocyte, activating the melanocyte. Palmaria palmata inhibits the release of SCF and therefore inhibits melanocyte activation; in addition, it inhibits Tyrosinase activity and the synthesis of the protein anchoring complex required for transfer of melanosomes to keratinocytes.
Ascorbic acid or vitamin C has been used for many years to control melanin synthesis. Newer stabilized derivatives of vitamin C, including magnesium ascorbyl phosphate, ascorbyl glucoside, and tetrahexyldecyl ascorbate are used to control melanogenesis. These derivatives can scavenge free radicals that cause erratic melanocyte activity, as well as act as antioxidants inhibiting oxidation steps along the biosynthetic pathway (i.e. DOPA to Dopaquinone) of melanin. They have also been shown to inhibit tyrosinase synthesis and activity.
Finally, the newest agents to fight melanin formation are the peptides. Oligopeptide-34 is a state-of-the-art peptide synthesized by an Asian pharmaceutical company that has been shown to decrease alpha-MSH activity and inhibit Tyrosinase activity. While the mechanism is not clearly understood, results indicate that it brightens skin, especially sun-induced hyperpigmentation, in half the time when compared to other brightening complexes.
Having Realistic Expectations
I always advise students that the first consideration in selecting which type of product to use on a client has to be first for their safety, then for the effectiveness of the product. Regardless of which brightening agents one selects for treating hyperpigmentation, the key thing to remember is that you must consistently use the product every morning and night for at least eight weeks before you will detect an improvement. Even if you could shut down all new melanin biosynthesis in the skin, remember it still takes about 45 days for the existing melanin to naturally be sloughed off. At the same time you are treating existing pigmentation, you must use a minimum SPF 15 on the skin whenever outdoors. Too often someone is religious about application of his or her pigment lightener/brightener products but then they forget to apply a sunscreen and within 24 hours the hyperpigmentation spots reappear.
Remember also the depth of the pigmentation will dictate the degree of effectiveness when treating hyperpigmentation. Dermal pigmentation is more difficult to treat than epidermal pigment. For obvious reasons, hormonally induced pigmentation and deeper post-inflammatory hyperpigmentation are often more difficult to treat than sun-induced pigmentation. And remember, you and your client should have realistic expectations as to the degree of lightening that you will achieve. It is highly unlikely that you will make a spot disappear but you can reduce its intensity and help to even-out the overall pigmentation of the skin. Not all hyperpigmentation spots will respond favorably – if 75 percent of your clients get significant lightening to hyperpigmentation that is considered a very good result.
Diana Howard Ph.D. has been in the cosmetic industry for 23 years. She earned her doctorate degree from The University of California Los Angeles where she specialized in plant biochemistry. Since then she has worked in research and development, as well as marketing for many of the major cosmetic companies. In addition, Howard was General Manager of the Leonard Drake Skin Care Centers where she developed the protocol required for opening and operating a successful skin care center. She has lectured extensively around the world, sharing her knowledge of hair and skin research, ingredient technology, and business skills for achieving ones full business potential in the skin and spa industry. She is currently Vice President of Technical Development for The International Dermal Institute and Dermalogica.
Want to read more?
Subscribe to one of our monthly plans to continue reading this article.