
Ultraviolet (UV) light from the sun is a major health hazard. Not only is it responsible for the greatest number of cancers of the skin, but it is a major cause of aging skin, as well as cataracts and related conditions. An understanding of exactly how UV light interacts with the skin’s components is critical to an appreciation of just how damaging UV rays can be.
The Photon
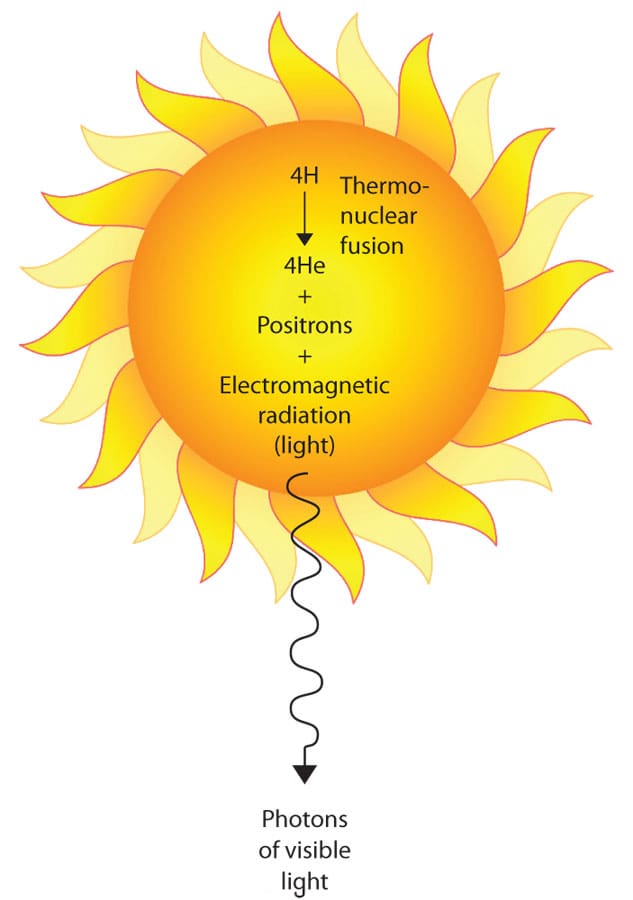
The key concept in understanding light energy is the photon. A photon is actually a very tiny particle that travels in a wave pattern from the sun to the earth. Each photon has a specific amount of energy, which scientists measure in several ways.
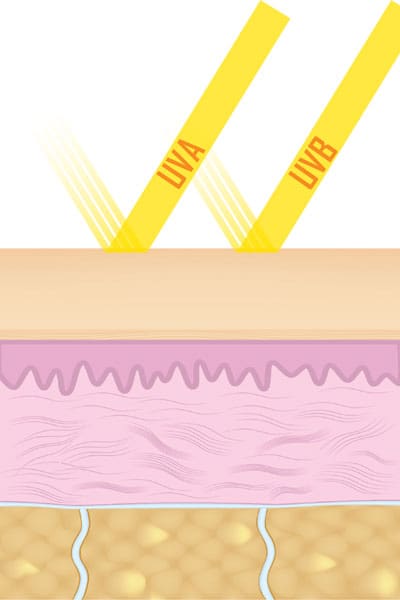
The most common way of measuring photon energy is in energy units known as joules. The other method commonly used is in electron volts, generally abbreviated eV. There is a relationship between the wavelength at which a photon travels and the amount of energy it carries. We call this an inverse relationship, because the shorter the wavelength, the greater is the amount of energy carried by the photon. For example, UV light designated as UVA travels at a wavelength of 315 to 400 nm, and is 3.94 eV to 3.10 eV, whereas UVB is 285 to 315 nm, and ranges from 3.94 to 4.43 eV. How does this energy relate to damage in the skin?
All biological structures are composed of molecules, which in turn are composed of atoms, which are held together by energy bonds. It is characteristic of each molecular structure that the bond energy is always specific for a particular type of bond. For example, a carbon-to-carbon bond, written as C-C, will be of a different energy than a carbon-to-oxygen bond, written as C-O. For light to have an effect on a chemical structure, it must be able unite with that compound. As an example, the major target in the skin for UV light is the DNA molecule, since it is so large and has so many components. When a ray of UVB light strikes a particular molecular structure in the epidermis (such as a purine, or pyrimidine base in the DNA) it will combine with that particular molecular structure in such a way that it will have one of three effects. Extra energy, when absorbed by a molecular structure, can be dissipated as heat. It may also be irradiated as light at a lower energy, a process known as fluorescence. Both methods will allow the molecule to return to its natural state when the extra energy disappears. The third method is more complex. The molecule will undergo a change in molecular structure to be able to accommodate the extra energy. This is not a good thing, for the change in structure may alter the molecule to where it becomes abnormal, and thus is essentially a different compound. We call this new compound an adduct. The body has a number of systems that are able to correct these abnormal molecules or eliminate them. As an example, often times the DNA molecule, when irradiated with UV light at certain energy levels, will chemically combine two adjacent bases-to-bases such as thymine. These two bases will become chemically linked together, side by side, thus forming a new compound known as a dimer. These abnormal bases are nonfunctional and cannot be reproduced by the body, so they have to be eliminated and replaced with a normal molecule of thymine.
As the sun’s energy enters the skin, it is absorbed by a large number of chemical compounds in the skin. The depth of penetration depends on the wavelength of the UV light striking the skin. UV light in the UVB range will affect primarily the tissues and cells in the epidermis, while the UVA range will interact more readily with the dermal structures.
You can take advantage of this terminology, UVA and UVB, to help you to remember the types of damage they cause. Associate UVB with basal cell carcinoma and UVA with aging.
While there may be considerable overlap between the effects of both UVA and UVB, as a rule, the energy in UVB is much stronger than UVA, so it creates a different kind of damage. Much of the damage involves alteration of DNA. Keep in mind, also, that the effect of the UV light will always depend on the amount of energy in the light, as well as the length of time an individual is exposed. There are a few practical things to remember. UV light exposure is highest between the hours of 10 a.m. and 2 p.m. The angle at which the sun strikes the earth is also critical, so that morning and evening sunrays are less potent, since they contain mostly infrared light. Anything that tends to scatter the sunlight will reduce its energy level, but does not completely eliminate the danger from UV light. A cloudy day, therefore, will offer you some protection, but not enough to allow you to stay out safely all day. Many a fisherman has learned this to his or her regret.
Vitamin D
People often ask about exposure to UV light and production of vitamin D. In the summer, UVB rays form a reaction in the skin cells that will produce vitamin D. A fair-skinned person exposed to midday sun for 10 minutes, with enough skin exposed (as in, wearing a swimsuit) will get enough UV radiation to produce about 10,000 international units of the vitamin. Unfortunately, if your skin type is a high Fitzpatrick number, you will not have enough to supply your needs for vitamin D from exposure to the sun. And, UVB rays cannot fully penetrate the atmosphere in the wintertime, due to the low angle of the winter sun. At the present time, the current United States government’s recommendation for quantities of vitamin D is believed, by many experts, to be too low. In general, the recommendation of 2,000 international units per day in the wintertime would be an effective dose, and also in the summer, if you are not a sun lover. Vitamin D is known to protect individuals against breast cancer, prostate and colon cancer, osteoporosis and depression. There is also evidence that adequate doses of vitamin D may be protective against heart disease.
How do we protect ourselves from the harmful effects of the sun? There are several ways to avoid sun damage. Obviously, staying out of the sun and/or wearing sensible protective clothing are two ways. For those individuals who want to enjoy the benefits of the sun, there are products that can afford some protection. It is important to understand that no product will give you 100 percent protection from UV light from the sun. There are two ways to reduce the sun’s UV light using commercial products. The first method is to employ physical blocks, such as afforded by zinc oxide or titanium dioxide. These inorganic compounds are now ground to nanometer size, which makes them invisible on the skin, eliminating the ugly white rub-out of the products. The nanoparticles, however, absorb the UV light and chemically produce free radicals. There are pulmonary studies which indicate that these free radicals are capable of reacting with DNA in the skin. Initial studies show that nanoparticles of zinc and titanium dioxide do not penetrate beyond the stratum corneum. For all intents and purposes, we can say these products appear to be safe, but the final word is not in. The many chemical sunscreens in use absorb the energy of the UV light as mentioned above, and can undergo chemical changes that produce potentially dangerous adducts. Recently, a product known as SolastayTM (ethylhexyl methoxycrylene, Hallstar) was originally designed for use in skin care and sun care products. It is a highly efficient singlet state quencher for Butyl Methoxydibenzoylmethane (BMDBM; Avobenzone) and Ethylhexyl Methoxycinnamate (OMC; Octinoxate). Singlet oxygen is the common name used for an electronically excited state of molecular oxygen (O2), which is less stable than the normal form but is capable of producing free radicals. When added to a sunscreen formula, SolastayTM is capable of stabilizing the chemical sunscreens to prevent them from becoming harmful adducts. The addition of this product should make the use of chemical sunscreens considerably safer.
When UV light passes through the skin, it is very important for the aesthetician to understand what is happening. Keep in mind that high energy of different frequencies contained in the waves all of UV light will interact with many of the complex proteins in the skin. Proteins, being composed of amino acids, by nature have a tendency to coil and cross-link to specific configurations that are critical to the functioning of a particular protein. Anything that alters this functional configuration will, in essence, denature the protein. Let us look at elastin, the very important protein that contributes to skin appearance, yet it makes up only one to two percent of the proteins in the skin. It is now known that UVA is capable of inducing changes in the fibroblasts of the skin, to induce them to produce more elastin. Most of the elastin produced in the body is produced in our youth, but the elastin produced in mature years tends to aggregate and form a less functional elastic material. Dermatologists have designated this condition as solar elastosis. It is one of the prime reasons that skin assumes a sagging, thin appearance with age. We can help to prevent these changes by the addition of both topical and systemic antioxidants.
DNA
Vitamin E and vitamin C are critical to the protection of our skin. The skin receives very little of the vitamin C that we ingest orally, so it is helpful if the vitamin C intake is increased when we venture out into the sun. The addition of lutein and lycopene as supplements, along with green tea, should prove helpful in avoiding sun damage. Keep in mind that ultimately the effect of the UV light is to damage the DNA. In response, the body has developed repair mechanisms known as “excision repair.” This is a system in which the body will repeatedly check the DNA for the presence of abnormal bases, that is adenine, thymine, cytosine and guanine. Frequently, UV light cross-links to adjacent bases as mentioned above, forming a dimer, which is not only nonfunctional but also cannot be reproduced. The body therefore will cut out this abnormal compound and replace it with a new functional base.
There is a disease in which an individual does not have the excision repair mechanism for DNA. It is known as xeroderma pigmentosa (XP) and it is characterized by multiple skin cancers. In extreme cases, all exposure to sunlight must be forbidden; essentially these are “Children of the Night.” Multiple basal cell carcinomas (basaliomas) and other skin malignancies frequently occur at a young age in those with XP. Actually, metastatic malignant melanoma and squamous cell carcinoma are the two most common causes of death in XP victims. This is an autosomal recessive disease, that is, both parents must carry the gene that transmits the disease. It is found most frequently in offspring who are the result of consanguinity (close blood relations). There is no known treatment, only prevention, by completely avoiding the sun.
We have come a long way in our knowledge of the sun’s impact on our health, and have seen the results of our skin’s response to exposure. Patti Pugliese, L.E. and daughter of Dr. Pugliese, says, “Apart from the wrinkles and spots, let us not forget the true wondrous function of our skin, the role it plays in our very survival. Our goal is that the children of the next century will know skin cancer only as an old-fashioned disease, like scurvy. And like the sailors eating limes to prevent scurvy, we know what to do. And when you know better, you do better.”


I blog often and I seriously thank you for your content.
This article has really peaked my interest. I am going to
take a note of your blog and keep checking for new information about once a week.
I opted in for your Feed too.
Leave a Reply
You must be logged in to post a comment.





















1 Comments